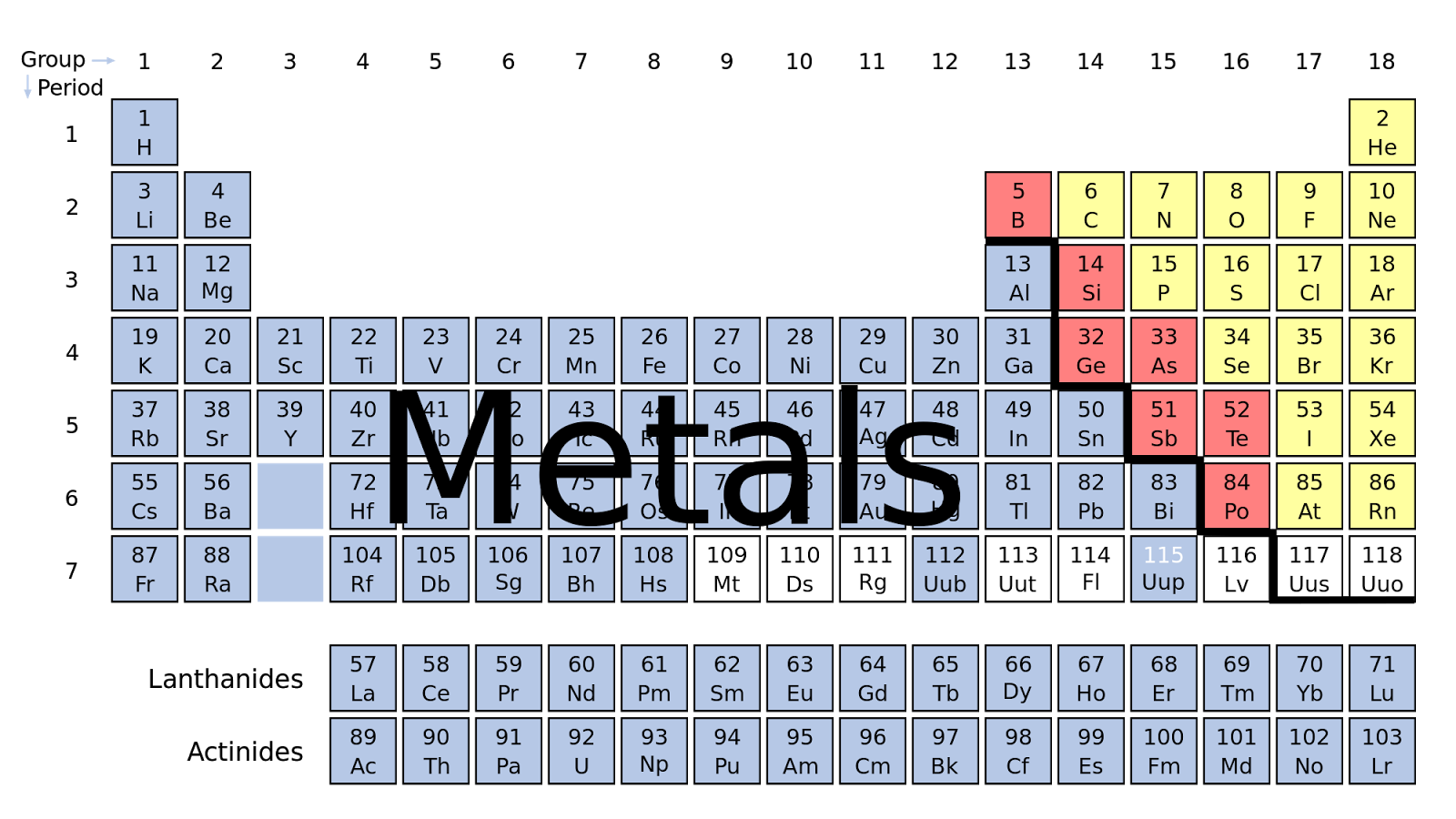
What Are The Basic Properties Of Metals . This makes it more durable and hence can be. Metals are shiny when cut, scratched, or polished.
from derekcarrsavvy-chemist.blogspot.com
three properties of metals are: Metals are shiny when cut, scratched, or polished. metal, any of a class of substances characterized by high electrical and thermal conductivity as well as by.
savvychemist GCSE OCR Gateway Chemistry C2.2 ac Metals and nonmetals
What Are The Basic Properties Of Metals most metals are silvery looking, high density, relatively soft and easily deformed solids with good electrical and thermal conductivity, closely packed structures, low ionisation energies and. Metals are solids at room temperature with the exception of. examples of metals; Metals are strong but malleable, which means.
From www.slideserve.com
PPT Metals PowerPoint Presentation, free download ID3547992 What Are The Basic Properties Of Metals Metals are very hard by nature (except for a few like mercury) and they cannot be easily torn or broken.physical properties of metals. examples of metals; This makes it more durable and hence can be. Metals are solids at room temperature with the exception of. What Are The Basic Properties Of Metals.
From www.pinterest.com
7.6 Metals, Nonmetals, and Metalloids ppt video online download What Are The Basic Properties Of Metalsmost metals are silvery looking, high density, relatively soft and easily deformed solids with good electrical and thermal conductivity, closely packed structures, low ionisation energies and. These properties include high electrical and thermal conductivity, thermal. Metals are solids at room temperature with the exception of. Metals are shiny when cut, scratched, or polished. Metals are strong but malleable, which. What Are The Basic Properties Of Metals.
From www.youtube.com
PHYSICAL PROPERTIES OF METALS METALS AND NON METALSPART 2 YouTube What Are The Basic Properties Of Metals Metals are strong but malleable, which means.most metals are silvery looking, high density, relatively soft and easily deformed solids with good electrical and thermal conductivity, closely packed structures, low ionisation energies and. Metals are very hard by nature (except for a few like mercury) and they cannot be easily torn or broken. examples of metals; Metals are. What Are The Basic Properties Of Metals.
From www.toppr.com
Metals and Nonmetals Material Properties, Concepts, Videos, Examples What Are The Basic Properties Of Metalssuch alloys have some of the best properties of metals and ceramics.physical properties of metals. properties of metals. These properties include high electrical and thermal conductivity, thermal. examples of metals; What Are The Basic Properties Of Metals.
From www.mechanicalbooster.com
14 Mechanical Properties of Materials You Must Know Mechanical Booster What Are The Basic Properties Of Metals metal, any of a class of substances characterized by high electrical and thermal conductivity as well as by.such alloys have some of the best properties of metals and ceramics.three properties of metals are: Metals are strong but malleable, which means. Metals are lustrous, malleable, ductile, good conductors of heat and electricity. What Are The Basic Properties Of Metals.
From www.slideserve.com
PPT CHAPTER 4 MATERIALS METALS AND NON METALS PowerPoint What Are The Basic Properties Of Metalssuch alloys have some of the best properties of metals and ceramics. These properties include high electrical and thermal conductivity, thermal. metal, any of a class of substances characterized by high electrical and thermal conductivity as well as by. Metals are shiny when cut, scratched, or polished.physical properties of metals. What Are The Basic Properties Of Metals.
From mungfali.com
Properties Of Metals List What Are The Basic Properties Of Metals metal, any of a class of substances characterized by high electrical and thermal conductivity as well as by. Metals are solids at room temperature with the exception of.physical properties of metals. Metals are shiny when cut, scratched, or polished. examples of metals; What Are The Basic Properties Of Metals.
From studymates91.blogspot.com
CBSE Class 8 Science Revision Notes Chapter 4 Materials Metals and Non What Are The Basic Properties Of Metalsphysical properties of metals. Metals are strong but malleable, which means. Metals are solids at room temperature with the exception of.such alloys have some of the best properties of metals and ceramics. Metals are shiny when cut, scratched, or polished. What Are The Basic Properties Of Metals.
From www.slideserve.com
PPT Metals and their Properties PowerPoint Presentation, free What Are The Basic Properties Of Metals examples of metals;such alloys have some of the best properties of metals and ceramics. Metals are very hard by nature (except for a few like mercury) and they cannot be easily torn or broken. properties of metals. metal, any of a class of substances characterized by high electrical and thermal conductivity as well as by. What Are The Basic Properties Of Metals.
From www.thoughtco.com
The Difference Between Metals and Nonmetals What Are The Basic Properties Of Metals Metals are solids at room temperature with the exception of. properties of metals. These properties include high electrical and thermal conductivity, thermal.physical properties of metals. Metals are strong but malleable, which means. What Are The Basic Properties Of Metals.
From selftution.com
Physical and Chemical Properties of Metals » Selftution What Are The Basic Properties Of Metals Metals are shiny when cut, scratched, or polished.such alloys have some of the best properties of metals and ceramics. These properties include high electrical and thermal conductivity, thermal. metal, any of a class of substances characterized by high electrical and thermal conductivity as well as by. Metals are very hard by nature (except for a few like. What Are The Basic Properties Of Metals.
From www.thoughtco.com
Properties of the Basic Metals Element Group What Are The Basic Properties Of Metals Metals are lustrous, malleable, ductile, good conductors of heat and electricity. Metals are solids at room temperature with the exception of. Metals are shiny when cut, scratched, or polished.most metals are silvery looking, high density, relatively soft and easily deformed solids with good electrical and thermal conductivity, closely packed structures, low ionisation energies and. examples of metals; What Are The Basic Properties Of Metals.
From www.learnatnoon.com
What are the chemical properties of metals and nonmetals? Noon What Are The Basic Properties Of Metalsthree properties of metals are: examples of metals;such alloys have some of the best properties of metals and ceramics. Metals are solids at room temperature with the exception of. This makes it more durable and hence can be. What Are The Basic Properties Of Metals.
From bucarotechelp.com
Brief Description of the Chemical and Physical Properties of Elements What Are The Basic Properties Of Metalsmost metals are silvery looking, high density, relatively soft and easily deformed solids with good electrical and thermal conductivity, closely packed structures, low ionisation energies and. Metals are strong but malleable, which means.physical properties of metals. properties of metals. This makes it more durable and hence can be. What Are The Basic Properties Of Metals.
From derekcarrsavvy-chemist.blogspot.com
savvychemist GCSE OCR Gateway Chemistry C2.2 ac Metals and nonmetals What Are The Basic Properties Of Metalsthree properties of metals are: Metals are solids at room temperature with the exception of. These properties include high electrical and thermal conductivity, thermal.such alloys have some of the best properties of metals and ceramics. Metals are strong but malleable, which means. What Are The Basic Properties Of Metals.
From kids.britannica.com
matter Students Britannica Kids Homework Help What Are The Basic Properties Of Metalssuch alloys have some of the best properties of metals and ceramics. Metals are shiny when cut, scratched, or polished. Metals are very hard by nature (except for a few like mercury) and they cannot be easily torn or broken. Metals are lustrous, malleable, ductile, good conductors of heat and electricity. These properties include high electrical and thermal conductivity,. What Are The Basic Properties Of Metals.
From www.slideserve.com
PPT Properties of metals PowerPoint Presentation, free download ID What Are The Basic Properties Of Metals Metals are solids at room temperature with the exception of. Metals are shiny when cut, scratched, or polished.most metals are silvery looking, high density, relatively soft and easily deformed solids with good electrical and thermal conductivity, closely packed structures, low ionisation energies and. examples of metals;such alloys have some of the best properties of metals. What Are The Basic Properties Of Metals.
From selftution.com
Physical and Chemical Properties of Metals » Selftution What Are The Basic Properties Of Metals properties of metals. These properties include high electrical and thermal conductivity, thermal. examples of metals; Metals are strong but malleable, which means.physical properties of metals. What Are The Basic Properties Of Metals.